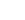CH + H
Gaz phase reaction (type: Bimolecular reactions)
Datasheet T(K) = 10-500
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
10-500 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2011-10-26 |
CH + H → C + H2
T(K): 10-500 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2011-10-26 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
10-500 | 5.12e-11 | 1.24E-10 | 2.60E-1 | 0.00E+0 | 3 | 14 | lognormal |
CH + H → C + H2
T(K) = 10-500 α = 1.24E-10 β = 2.60E-1 γ = 0.00E+0 F 0 = 3 g = 14 Type uncert: lognormal Method: Reviews and Evaluations |
|
Method: Reviews and Evaluations
|
|||||||||
Van Harrevelt, R. et al (2002) T(K) = 50-500
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
50-500 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2009-07-16 |
CH + H → C + H2
T(K): 50-500 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2009-07-16 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
50-500 | 5.12e-11 | 1.24E-10 | 2.60E-1 | 0.00E+0 | 2 | 0 | loguniform |
CH + H → C + H2
T(K) = 50-500 α = 1.24E-10 β = 2.60E-1 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: loguniform Method: Calculations |
|
Method: Calculations
|
|||||||||
Becker, K. H. et al (1989) T(K) = 300-300
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
300-300 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2009-07-16 |
CH + H → C + H2
T(K): 300-300 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2009-07-16 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
300-300 | 1.40e-11 | 1.40E-11 | 0.00E+0 | 0.00E+0 | 0 | 0 | lognormal |
CH + H → C + H2
T(K) = 300-300 α = 1.40E-11 β = 0.00E+0 γ = 0.00E+0 F 0 = 0 g = 0 Type uncert: lognormal Method: Measurements |
|
Method: Measurements
|
|||||||||
Grebe, J. et al (1982) T(K) = 298-298
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
298-298 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2010-05-28 |
CH + H → C + H2
T(K): 298-298 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2010-05-28 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
298-298 | 4.90e-11 | 4.90E-11 | 0.00E+0 | 0.00E+0 | 0 | 0 | lognormal |
CH + H → C + H2
T(K) = 298-298 α = 4.90E-11 β = 0.00E+0 γ = 0.00E+0 F 0 = 0 g = 0 Type uncert: lognormal Method: Measurements |
|
Method: Measurements
|
|||||||||
Other database : OSU T(K) = 10-280
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
10-280 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2009-03-27 |
CH + H → C + H2
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2009-03-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
10-280 | 7.41e-12 | 2.70E-11 | 3.80E-1 | 0.00E+0 | 2 | 0 | lognormal |
CH + H → C + H2
T(K) = 10-280 α = 2.70E-11 β = 3.80E-1 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
| Reference | |||||||
|---|---|---|---|---|---|---|---|
| Other database : OSU | |||||||
|
Origin: Other database : OSU
Authors: Volume,Page: Year: DOI: - Download citation: |
|||||||
Hebrard, E. et al (2009) T(K) = 50-200
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
50-200 | Modified Arrhenius equation |
ΔrH0 = -102.6
Δ rH298 = -97.128 |
2012-07-27 |
CH + H → C + H2
T(K): 50-200 Formula: Modified Arrhenius equation Δ rH0 = -102.6 Δ rH298 = -97.128 2012-07-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH + H
→
C + H2
|
50-200 | 4.39e-14 | 1.31E-10 | 0.00E+0 | 8.00E+1 | 10 | 100 | lognormal |
CH + H → C + H2
T(K) = 50-200 α = 1.31E-10 β = 0.00E+0 γ = 8.00E+1 F 0 = 10 g = 100 Type uncert: lognormal Method: Reviews and Evaluations Description: Taken from Harding et al. [1993]. Used in Hébrard et al. [2009] |
|
Method: Reviews and Evaluations
Description: Taken from Harding et al. [1993]. Used in Hébrard et al. [2009] |
|||||||||