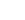C3 + N
Gaz phase reaction (type: Bimolecular reactions)
Datasheet T(K) = 10-298
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
C3 + N
→
C2 + CN
|
10-298 | Modified Arrhenius equation |
ΔrH0 = -38.995
Δ rH298 = -29.226 |
2009-05-11 |
C3 + N → C2 + CN
T(K): 10-298 Formula: Modified Arrhenius equation Δ rH0 = -38.995 Δ rH298 = -29.226 2009-05-11 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
C3 + N
→
C2 + CN
|
10-298 | 0.00e+0 | 0.00E+0 | 0.00E+0 | 0.00E+0 | 0 | 0 | lognormal |
C3 + N → C2 + CN
T(K) = 10-298 α = 0.00E+0 β = 0.00E+0 γ = 0.00E+0 F 0 = 0 g = 0 Type uncert: lognormal Method: Estimation Description: This reaction seems slow at 298 K and very slow at 10 K. Although the reaction is not very important for interstellar chemistry, we have set its rate coefficient to 0 in order to avoid any effect. |
|
Method: Estimation
Description: This reaction seems slow at 298 K and very slow at 10 K. Although the reaction is not very important for interstellar chemistry, we have set its rate coefficient to 0 in order to avoid any effect. |
|||||||||
Other database : OSU T(K) = 10-280
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
C3 + N
→
C2 + CN
|
10-280 | Modified Arrhenius equation |
ΔrH0 = -38.995
Δ rH298 = -29.226 |
2009-03-27 |
C3 + N → C2 + CN
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = -38.995 Δ rH298 = -29.226 2009-03-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
C3 + N
→
C2 + CN
|
10-280 | 1.00e-13 | 1.00E-13 | 0.00E+0 | 0.00E+0 | 2 | 0 | lognormal |
C3 + N → C2 + CN
T(K) = 10-280 α = 1.00E-13 β = 0.00E+0 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
| Reference | |||||||
|---|---|---|---|---|---|---|---|
| Other database : OSU | |||||||
|
Origin: Other database : OSU
Authors: Volume,Page: Year: DOI: - Download citation: |
|||||||