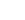CH3CN + H3O+
Gaz phase reaction (type: Bimolecular reactions)
Woon, D. E. et al (2009) T(K) = 10-800
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
10-800 | ionpol1 |
ΔrH0 = N/A
Δ rH298 = N/A |
2011-10-17 |
CH3CN + H3O+ → H2O + CH3CNH+
T(K): 10-800 Formula: ionpol1 Δ rH0 = N/A Δ rH298 = N/A 2011-10-17 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
10-800 | 2.40e-8 | 1.00E+0 | 1.35E-9 | 6.58E+0 | 2 | 0 | lognormal |
CH3CN + H3O+ → H2O + CH3CNH+
T(K) = 10-800 α = 1.00E+0 β = 1.35E-9 γ = 6.58E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Calculations Description: Rate coefficients for ion-polar reactions are computed by two different formula depending on the temperature. Note that the formula are different from the Arrhenius-Kooij form used for the other bimolecular reactions. Alpha represents the branching ratio of the channel and beta the langevin rate. Some details can also be found in Wakelam et al. (2010, Space Science Reviews, 156, 13) |
|
Method: Calculations
Description: Rate coefficients for ion-polar reactions are computed by two different formula depending on the temperature. Note that the formula are different from the Arrhenius-Kooij form used for the other bimolecular reactions. Alpha represents the branching ratio of the channel and beta the langevin rate. Some details can also be found in Wakelam et al. (2010, Space Science Reviews, 156, 13) |
|||||||||
Anicich, V. G. (2003) T(K) = 300-300
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
300-300 | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2012-07-30 |
CH3CN + H3O+ → H2O + CH3CNH+
T(K): 300-300 Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2012-07-30 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
300-300 | 4.50e-9 | 4.50E-9 | 0.00E+0 | 0.00E+0 | 1.15 | 0 | lognormal |
CH3CN + H3O+ → H2O + CH3CNH+
T(K) = 300-300 α = 4.50E-9 β = 0.00E+0 γ = 0.00E+0 F 0 = 1.15 g = 0 Type uncert: lognormal Method: Reviews and Evaluations Description: Taken from Anicich, J. Phys. Chem. Ref. Data 22, 1469-1569 (1993) |
|
Method: Reviews and Evaluations
Description: Taken from Anicich, J. Phys. Chem. Ref. Data 22, 1469-1569 (1993) |
|||||||||
Other database : OSU T(K) = 10-280
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
10-280 | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-03-27 |
CH3CN + H3O+ → H2O + CH3CNH+
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-03-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
CH3CN + H3O+
→
H2O + CH3CNH+
|
10-280 | 2.31e-8 | 4.22E-9 | -5.00E-1 | 0.00E+0 | 1.25 | 0 | lognormal |
CH3CN + H3O+ → H2O + CH3CNH+
T(K) = 10-280 α = 4.22E-9 β = -5.00E-1 γ = 0.00E+0 F 0 = 1.25 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
| Reference | |||||||
|---|---|---|---|---|---|---|---|
| Other database : OSU | |||||||
|
Origin: Other database : OSU
Authors: Volume,Page: Year: DOI: - Download citation: |
|||||||