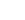HCNH+ + e-
Gaz phase reaction (type: Electronic recombination and attachment)
Datasheet T(K) = 10-1000
Show/Hide those values
| Channels | ||||||
|---|---|---|---|---|---|---|
|
HCNH+ + e-
→
H + H + CN
|
10-1000 | 32 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-10-22 |
HCNH+ + e- → H + H + CN
T(K): 10-1000 Branching ratio: 32 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-10-22 |
|
HCNH+ + e-
→
H + HCN
|
10-1000 | 34 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-10-22 |
HCNH+ + e- → H + HCN
T(K): 10-1000 Branching ratio: 34 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-10-22 |
|
HCNH+ + e-
→
H + HNC
|
10-1000 | 34 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-10-22 |
HCNH+ + e- → H + HNC
T(K): 10-1000 Branching ratio: 34 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-10-22 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
HCNH+ + e-
→
H + H + CN
|
10-1000 | 8.26e-7 | 9.06E-8 | -6.50E-1 | 0.00E+0 | 2 | 0 | lognormal |
HCNH+ + e- → H + H + CN
T(K) = 10-1000 α = 9.06E-8 β = -6.50E-1 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Reviews and Evaluations Description: See datasheet - review in the context of the ISSI group |
|
Method: Reviews and Evaluations
Description: See datasheet - review in the context of the ISSI group |
|||||||||
|
HCNH+ + e-
→
H + HCN
|
10-1000 | 8.78e-7 | 9.62E-8 | -6.50E-1 | 0.00E+0 | 2 | 0 | lognormal |
HCNH+ + e- → H + HCN
T(K) = 10-1000 α = 9.62E-8 β = -6.50E-1 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Reviews and Evaluations Description: See datasheet - review in the context of the ISSI group |
|
Method: Reviews and Evaluations
Description: See datasheet - review in the context of the ISSI group |
|||||||||
|
HCNH+ + e-
→
H + HNC
|
10-1000 | 8.78e-7 | 9.62E-8 | -6.50E-1 | 0.00E+0 | 2 | 0 | lognormal |
HCNH+ + e- → H + HNC
T(K) = 10-1000 α = 9.62E-8 β = -6.50E-1 γ = 0.00E+0 F 0 = 2 g = 0 Type uncert: lognormal Method: Reviews and Evaluations Description: See datasheet - review in the context of the ISSI group |
|
Method: Reviews and Evaluations
Description: See datasheet - review in the context of the ISSI group |
|||||||||
Other database : OSU T(K) = 10-280
Show/Hide those values
| Channels | ||||||
|---|---|---|---|---|---|---|
|
HCNH+ + e-
→
H + H + CN
|
10-280 | 20 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-03-27 |
HCNH+ + e- → H + H + CN
T(K): 10-280 Branching ratio: 20 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-03-27 |
|
HCNH+ + e-
→
H + HCN
|
10-280 | 40 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-03-27 |
HCNH+ + e- → H + HCN
T(K): 10-280 Branching ratio: 40 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-03-27 |
|
HCNH+ + e-
→
H + HNC
|
10-280 | 40 % | Modified Arrhenius equation |
ΔrH0 = N/A
Δ rH298 = N/A |
2009-03-27 |
HCNH+ + e- → H + HNC
T(K): 10-280 Branching ratio: 40 % Formula: Modified Arrhenius equation Δ rH0 = N/A Δ rH298 = N/A 2009-03-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
HCNH+ + e-
→
H + H + CN
|
10-280 | 8.39e-7 | 9.20E-8 | -6.50E-1 | 0.00E+0 | 1.25 | 0 | lognormal |
HCNH+ + e- → H + H + CN
T(K) = 10-280 α = 9.20E-8 β = -6.50E-1 γ = 0.00E+0 F 0 = 1.25 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
|
HCNH+ + e-
→
H + HCN
|
10-280 | 1.69e-6 | 1.85E-7 | -6.50E-1 | 0.00E+0 | 1.25 | 0 | lognormal |
HCNH+ + e- → H + HCN
T(K) = 10-280 α = 1.85E-7 β = -6.50E-1 γ = 0.00E+0 F 0 = 1.25 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
|
HCNH+ + e-
→
H + HNC
|
10-280 | 1.69e-6 | 1.85E-7 | -6.50E-1 | 0.00E+0 | 1.25 | 0 | lognormal |
HCNH+ + e- → H + HNC
T(K) = 10-280 α = 1.85E-7 β = -6.50E-1 γ = 0.00E+0 F 0 = 1.25 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
| Reference | |||||||
|---|---|---|---|---|---|---|---|
| Other database : OSU | |||||||
|
Origin: Other database : OSU
Authors: Volume,Page: Year: DOI: - Download citation: |
|||||||