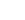C + NH2
Gaz phase reaction (type: Bimolecular reactions)
Herbst, E. et al (2000) T(K) = 10-300
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
C + NH2
→
H + HNC
|
10-300 | Modified Arrhenius equation |
ΔrH0 = -495.306
Δ rH298 = -492.964 |
2011-01-19 |
C + NH2 → H + HNC
T(K): 10-300 Formula: Modified Arrhenius equation Δ rH0 = -495.306 Δ rH298 = -492.964 2011-01-19 |
|
C + NH2
→
H + HCN
|
10-300 | Modified Arrhenius equation |
ΔrH0 = -557.16
Δ rH298 = -555.073 |
2011-01-19 |
C + NH2 → H + HCN
T(K): 10-300 Formula: Modified Arrhenius equation Δ rH0 = -557.16 Δ rH298 = -555.073 2011-01-19 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
C + NH2
→
H + HNC
|
10-300 | 1.08e-10 | 3.00E-11 | -2.00E-1 | -6.00E+0 | 1.5 | 0 | lognormal |
C + NH2 → H + HNC
T(K) = 10-300 α = 3.00E-11 β = -2.00E-1 γ = -6.00E+0 F 0 = 1.5 g = 0 Type uncert: lognormal Method: Calculations Description: Better fit of the data calculated by Herbst et al. (2000). HCN/HNC products are 50%/50%. |
|
Method: Calculations
Description: Better fit of the data calculated by Herbst et al. (2000). HCN/HNC products are 50%/50%. |
|||||||||
|
C + NH2
→
H + HCN
|
10-300 | 1.08e-10 | 3.00E-11 | -2.00E-1 | -6.00E+0 | 1.5 | 0 | lognormal |
C + NH2 → H + HCN
T(K) = 10-300 α = 3.00E-11 β = -2.00E-1 γ = -6.00E+0 F 0 = 1.5 g = 0 Type uncert: lognormal Method: Calculations Description: Better fit of the data calculated by Herbst et al. (2000). HCN/HNC products are 50%/50%. |
|
Method: Calculations
Description: Better fit of the data calculated by Herbst et al. (2000). HCN/HNC products are 50%/50%. |
|||||||||
Mayer, S.W. et al (1967) T(K) = 10-300
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
C + NH2
→
CH + NH
|
10-300 | Modified Arrhenius equation |
ΔrH0 = 48.39
Δ rH298 = 51.722 |
2011-01-19 |
C + NH2 → CH + NH
T(K): 10-300 Formula: Modified Arrhenius equation Δ rH0 = 48.39 Δ rH298 = 51.722 2011-01-19 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
C + NH2
→
CH + NH
|
10-300 | 0.00e+0 | 9.61E-13 | 0.00E+0 | 1.05E+4 | 10 | 0 | lognormal |
C + NH2 → CH + NH
T(K) = 10-300 α = 9.61E-13 β = 0.00E+0 γ = 1.05E+4 F 0 = 10 g = 0 Type uncert: lognormal Method: Calculations Description: Computation of high-temperature rate constants for bimolecular reactions of combustion products. It's not a reliable value. |
|
Method: Calculations
Description: Computation of high-temperature rate constants for bimolecular reactions of combustion products. It's not a reliable value. |
|||||||||
Other database : OSU T(K) = 10-280
Show/Hide those values
| Channels | |||||
|---|---|---|---|---|---|
|
C + NH2
→
CH + NH
|
10-280 | Modified Arrhenius equation |
ΔrH0 = 48.39
Δ rH298 = 51.722 |
2009-03-27 |
C + NH2 → CH + NH
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = 48.39 Δ rH298 = 51.722 2009-03-27 |
|
C + NH2
→
H + HNC
|
10-280 | Modified Arrhenius equation |
ΔrH0 = -495.306
Δ rH298 = -492.964 |
2009-03-27 |
C + NH2 → H + HNC
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = -495.306 Δ rH298 = -492.964 2009-03-27 |
|
C + NH2
→
H + HCN
|
10-280 | Modified Arrhenius equation |
ΔrH0 = -557.16
Δ rH298 = -555.073 |
2009-03-27 |
C + NH2 → H + HCN
T(K): 10-280 Formula: Modified Arrhenius equation Δ rH0 = -557.16 Δ rH298 = -555.073 2009-03-27 |
Show details
See/hide publications
|
k(
)
cm3s-1 |
Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
C + NH2
→
CH + NH
|
10-280 | 0.00e+0 | 9.61E-13 | 0.00E+0 | 1.05E+4 | 2 | 0 | lognormal |
C + NH2 → CH + NH
T(K) = 10-280 α = 9.61E-13 β = 0.00E+0 γ = 1.05E+4 F 0 = 2 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
|
C + NH2
→
H + HNC
|
10-280 | 1.16e-10 | 3.40E-11 | -3.60E-1 | 0.00E+0 | 1.5 | 0 | lognormal |
C + NH2 → H + HNC
T(K) = 10-280 α = 3.40E-11 β = -3.60E-1 γ = 0.00E+0 F 0 = 1.5 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
|
C + NH2
→
H + HCN
|
10-280 | 1.16e-10 | 3.40E-11 | -3.60E-1 | 0.00E+0 | 1.5 | 0 | lognormal |
C + NH2 → H + HCN
T(K) = 10-280 α = 3.40E-11 β = -3.60E-1 γ = 0.00E+0 F 0 = 1.5 g = 0 Type uncert: lognormal Method: Estimation |
|
Method: Estimation
|
|||||||||
| Reference | |||||||
|---|---|---|---|---|---|---|---|
| Other database : OSU | |||||||
|
Origin: Other database : OSU
Authors: Volume,Page: Year: DOI: - Download citation: |
|||||||